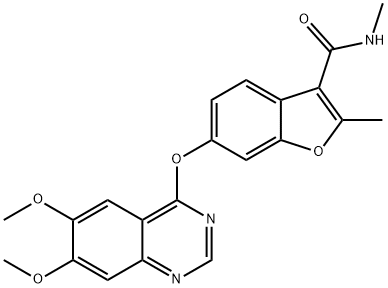
Fruquintinib API
| CAS No. | 1194506-26-7 |
| Therapeutic Category | Anti-Cancer/ Oncology |
| Technology | Synthetic |
| Dosage Form | Oral Solids |
| Innovator Brand | FRUZAQLA(USA) |
| Registration Status | Technical Package |
| Polymorph | Novel form |
| GMP | FDA(USA)/NMPA(China)/MFDS(Korea) approved |
| EHS | ISO 14001 & 45001 certified |
Product Description
- Grade: Active Pharmaceutical Ingredient(API)
- Chemical Name: 6-(6,7-dimethoxyquinazolin-4-yloxy)-N,2-dimethylbenzofuran-3-carboxamide;6-[(6,7-Dimethoxy-4-quinazolinyl)oxy]-N,2-dimethyl-3-benzofurancarboxamide
- Molecular Formula: C21H19N3O5
- Molecular Weight: 393.39
- Specification: Enterprise Standard established according to ChP/USP/EP
- Appearance: Powder
- Total impurities: not more than 0.5%
- Purity: not less than 99%
- Residual Solvents: fully comply with ICH Q3C
- Mutagenic impurities: fully comply with ICH M8
- Nitrosamine assessment: available
- Particle size: regular grade or milling/sieving according to customer’s requirement.
- Storage: Room temperature
- Production capacity: Commercial
- Standard Package: 1kg/bag, 5kg/bag, or according to the customer’s requirement
Applications of Fruquintinib API
- FRUZAQLA(Fruquintinib API), the first targeted therapy for mCRC regardless of mutation status approved in nearly a decade-is an oral medicine for adults with metastatic colorectal cancer (mCRC) that can be taken after chemotherapy and certain anti-cancer medicines to help people live longer.
- For adults with metastatic colorectal cancer (mCRC), FRUZAQLA is an innovative and convenient oral treatment with proven survival results that can be taken after chemotherapy and other anti-cancer medications.
- FRUZAQLA is a targeted therapy for people living with previously treated mCRC. FRUZAQLA is not chemotherapy
Why Choose Us as Your Fruquintinib API Manufacturer?
- Qingmu’s factory is established according to ICH/USFDA/EU/JAPAN/China regulations and current GMP, totally complies with environmental law in China, no risk to remove factory. A new API factory is under design and is predicted to be put into use in 2025.
- Qingmu’s team has rich experience in patent challenges on crystalline form & synthesis processes and also synthetic route development and scale-up & quality research.
- As a Fruquintinib manufacturer, Qingmu’s lab is equipped with HPLC, GC, ICP-MS, GC-MS, CAD, microbalance, Malvern particle analyzer, etc. Qingmu passed the site inspection from NMPA(China), FDA(USA) and MFDS(Korea) and also customer audits from Europe, USA and Japan, etc.
- Qingmu successfully exported products to more than 40 countries including Japan, USA, Germany, Spain, the Netherlands, Russia, South Korea, and Brazil. We are always ready to provide customers with the best quality products and services.